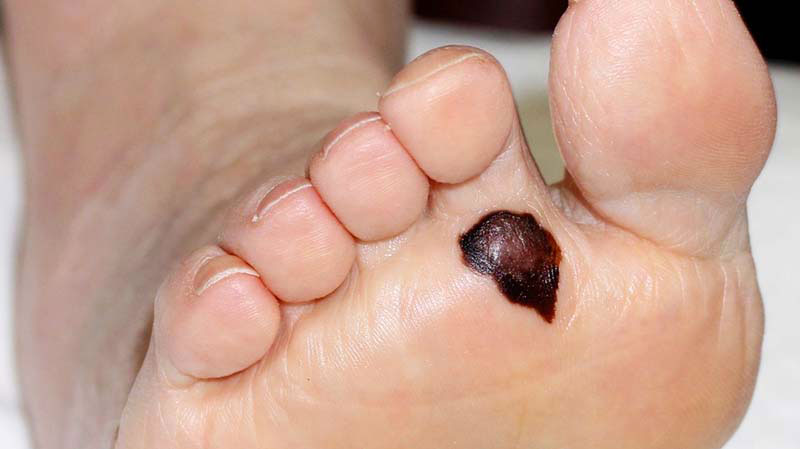
Using whole genome sequencing technologies to identify the genetic mutations that cause melanoma, so treatment can be personalised.
Melanomas develop following the accumulation of genetic mutations that enable the cells to grow uncontrollably. The Australian Melanoma Genome Project is using cutting-edge whole genome sequencing technologies to identify the common genetic mutations that cause melanoma so that doctors can better personalise its treatment.
The project has made major progress by comparing the genetic code of normal cells and melanoma tumours and identifying all the mutations present in nearly 500 Australian melanoma patients. It is the largest melanoma research effort ever undertaken in this country, with a national coalition of over 50 researchers from Melanoma Institute Australia, The University of Sydney, Royal Prince Alfred Hospital, Westmead Institute for Medical Research and the QIMR Berghofer Medical Research Institute working together.
Purpose
- Fully characterise all the genomic alterations in over 500 melanomas patients (currently 468 melanomas sequenced).
- Identify mutagenic processes and carcinogens responsible for the development of a range of melanomas.
- Discover new drug targets and better select patients for current therapeutics.
Highlights so far
Whole-genome landscapes of major melanoma subtypes
The study presents the whole genome sequencing data from the first 183 melanoma patients. The study highlighted some major difference in the types of genetic mutations, the affected genes and the carcinogens that drive melanomas that arise in sun exposed skin compared to those have arisen in mucosal surfaces and on the palms and soled of the hands and feet:
- Identifying drugs for patients:Acral and mucosal melanomas are dominated by large scale genetic changes that may offer new druggable targets to treat patients.
- Novel findings of the causes of a range of melanomas:The carcinogenic process or factors that cause a melanoma can be identified from the types of genomic mutations in a patient’s tumour. The study identified new mutation- causing processes driving melanoma development and found major differences in the causes of melanomas affecting sun exposed compared to those involving sun shielded sites. Original article: Hayward, N.K. et al. Whole-genome landscapes of major melanoma subtypes. 03 May 2017. doi: 10.1038/nature22071. [Epub ahead of print]
Public release of genomic data on freely available platforms
Our Melanoma Genome Project team recently played a crucial role in a global study that discovered cutaneous melanoma has four distinct subtypes. The breakthrough helps pave the way for more tailored individual, ‘personalised’ medicine for melanoma patients. Samples from the MIA BioSpecimen Bank made up almost one third of the global contributions to ‘The Cancer Genome Atlas’ (TCGA) melanoma project, a five year study of tumours from over 300 patients.
In addition, the Melanoma Genome Project was endorsed by the International Cancer Genome Consortium early in 2015. All of the data that has been generated by the project has been uploaded to the ICGC data portal to make it available for researchers, clinicians or the general public to interrogate thereby assisting the global cancer research community to accelerate progress to cure melanoma.
Original article: Cancer Genome Atlas Network. Genomic classification of cutaneous melanoma. Cell 161, 1681–1696 (2015).
Unexpected UVR and Non-UVR mutation burden in some acral and cutaneous melanomas
Analysis of the first 183 melanoma patients found sun exposure induced mutations in a two melanomas involving subungual (finger nail) region and one on the sole of the foot, regions generally considered to be protected from sun related mutations.
Original article: Rawson R.V., et al. Unexpected UVR and non-UVR mutation burden in some acral and cutaneous melanomas. Lab Invest,97 (2), 130-145 (2017)
Mutation load in melanoma is affected by MC1R genotype
Patients who harbour a germline (inheritable) mutation to the MC1R gene display increased sensitivity to sun light induced mutations.
Original article: Johansson P.A., et al., Mutation load in melanoma is affected by MC1R genotype. Pigment Cell Melanoma Res., 30: 255–258 (2017)
Best practices for sample preparation, sequencing and data analysis in clinical melanoma samples
Study outlined lessons learnt from the large-scale Australian Melanoma Genome Sequencing Project.
Clinical utility of whole exome sequencing in routine archival melanoma biopsies
This study assessed the accuracy of whole exome sequencing on routine archival formalin fixed melanoma biopsies.
Current projects
- Genomic determinants of melanoma patient response to immune checkpoint inhibitors
- Genomic landscapes of mucosal and acral melanomas
- Youth melanomas – Mutations and mutational processes responsible for melanoma development in young Australians (<30yrs old)
Funding
The Australian Melanoma Genome Project was funded directly by Melanoma Institute Australia, the NSW Ministry of Health, Cancer Council NSW and the Australian Government through Bioplatforms Australia.
The project has been enabled by Program Grants and Fellowships of the National Health and Medical Research Council, Translational Program Grants of Cancer Institute NSW, and infrastructure of the University of Sydney, Bioplatforms Australia, Westmead Institute for Medical Research, NSW Health Pathology, Royal Prince Alfred Hospital, Peter MacCallum Cancer Institute, The Olivia Newton-John Cancer Research Institute and Melanoma Institute Australia.
Project leads
From Melanoma Institute Australia:
- Prof Graham Mann– Co-Principal Investigator
- Prof Richard Scolyer– Co-Principal Investigator
- Dr James Wilmott– Project manager
From QIMR Berghofer Medical Research Institute:
- Prof Nick Hayward– Co-Principal Investigator
- Dr Nicola Waddell
- Dr Peter Johansson
- Dr John Pearson